RaDaR ST for Head & Neck Cancer
Closing the Surveillance Gap in HPV-Negative Head & Neck Cancer
HPV-negative head and neck squamous cell carcinoma (HNSCC) presents a significant clinical challenge, with many patients developing disease recurrence and fewer than 50% surviving beyond five years. HPV-negative patients face worse outcomes compared to their HPV-positive counterparts, and unlike HPV-positive disease, reliable biomarkers for therapy planning and monitoring treatment response do not exist for this population.
Current head and neck cancer adjuvant therapy monitoring and surveillance cannot answer two critical questions:
- Did surgery remove all the cancer?
- Are there ways to identify disease before clinical or radiographic signs appear?
This gap in monitoring leaves clinicians and patients without the tools needed to detect recurrence early when intervention may be most effective.
Earlier detection to support salvage treatment success
RaDaR ST delivers unprecedented sensitivity in detecting HPV-negative HNSCC recurrence, achieving 100% detection of relapsed patients by identifying ctDNA at ultra-low levels of 5ppm—ensuring no patient with recurrent disease goes undetected during the critical surveillance window.
Post-surgical ctDNA monitoring identifies disease relapse a median of 154 days (over 5 months) before clinical confirmation, providing crucial lead time when salvage surgery and radiation are most likely to succeed and before symptoms impact quality of life.
This sensitivity advantage is essential, as nearly one-third of positive samples had variant allele frequencies below 0.01%. It demonstrates standard sensitivity assays may miss a significant proportion of HNSCC relapse leaving patients to present with more advance disease.
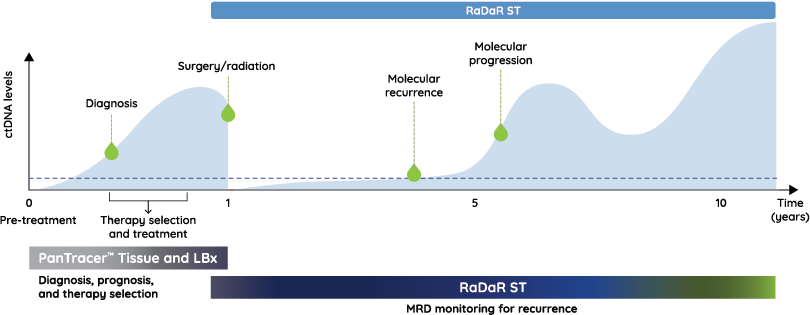
Head and neck cancer care continuum
From diagnosis through surveillance: delivering timely, actionable insights so that every treatment decision is as personal as the patients themselves.
Case study: When salvage treatment success rates are highest
When a 60-year-old man noticed a growing lump on his neck, he went to his doctor for evaluation. His medical history included 30 years of smoking and alcohol use, and his physical examination revealed a large, firm mass on the left side. A subsequent biopsy confirmed p16-negative squamous cell carcinoma. Imaging showed the cancer was still localized, and he underwent surgery to remove the mass.
Four weeks post-surgery, despite clear resection margins, RaDaR ST testing detected ctDNA in his blood. Additional surgery was performed within days, and his postoperative ctDNA levels dropped to undetectable. The patient continued regular monitoring with both imaging and RaDaR ST every 3 months. The patient's ctDNA appeared 9 months after surgery but imaging did not detect it until 6 months after RaDaR ST. The residual disease was identified by RaDaR ST months before imaging.*
*Data on file
References
Flach S, Howarth K, Hackinger S, et al. Liquid BIOpsy for MiNimal RESidual DiSease Detection in Head and Neck Squamous Cell Carcinoma (LIONESS)-a personalised circulating tumour DNA analysis in head and neck squamous cell carcinoma. Br J Cancer. 2022;126(8):1186-1195. doi:10.1038/s41416-022-01716-7