RaDaR ST for Breast Cancer
Address HR+/HER2- breast cancer long-term recurrence risk months before conventional methods
Hormone receptor (HR+) positive/HER2 negative tumors represent nearly 70% of breast cancer diagnoses and pose a unique clinical challenge. Unlike other breast cancer subtypes, late recurrences are common in patients with HR+/HER2- tumors, with over half of metastatic recurrences occurring 5+ years post-initial diagnosis. The risk of recurrence remains high for these patients for at least 20 years after curative intent treatment. This creates a significant unmet need, as current methods cannot detect breast cancer recurrence until tumors present on scans—missing the critical intervention window when disease burden is minimal and treatment is most likely to succeed.
RaDaR ST provides over a year's advance warning of distant metastasis, opening the door to life-changing interventions.
The test detects early evidence of recurrence, well before the tumor becomes metastatic, detecting 100% of distant metastatic recurrences in a 3.9-year study of 83 patients, with a remarkable median lead time of 12.4 months before clinical detection—providing an unprecedented window to intervene when disease burden is minimal and treatment may be most effective.1 For high-risk HR+/HER2- early-stage breast cancer patients, RaDaR ST extends vigilance beyond the early years to the long-term survival window by enabling continuous molecular surveillance in the late adjuvant setting, addressing the persistent threat of late recurrence that imaging alone may miss until it's too late.
Additionally, when endocrine-responsive biomarkers indicate high recurrence risk despite anti-estrogen therapy, integrating RaDaR ST testing can refine treatment decisions by detecting and monitoring circulating tumor DNA for more comprehensive risk assessment.
Breast cancer care continuum
From diagnosis through surveillance: delivering timely, actionable insights so that every treatment decision is as personal as the patients themselves.
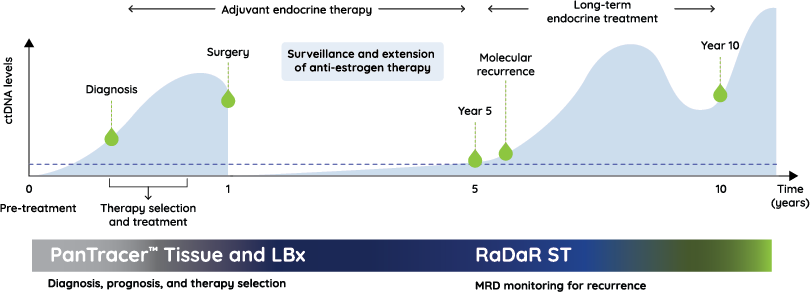
How RaDaR ST impacts the cancer care continuum
Case Study: Early detection changes the paradigm
A 52-year-old woman, diagnosed with Stage II ER+/PR+/HER2- breast cancer, completed her treatment successfully and showed no evidence of disease for 7 years. Then, during a routine 7-year surveillance, RaDaR ST detected ctDNA in her blood, even though imaging showed nothing unusual.
Her care team responded by scheduling more frequent monitoring, and 3 months later, the intensified imaging found a single bone lesion. She was quickly treated with targeted, local radiation and systemic therapy. The ctDNA cleared, and 18 months later, the patient remains on therapy with no signs of progression.*
*Data on file
Case study: Recurrence monitoring provides early warning
When a 52-year-old woman noticed swelling, pain, and itchiness in her left breast, her family physician ordered imaging that revealed resectable stage III breast cancer with 3 involved lymph nodes, but no distant metastases. Given her high risk for recurrence, she underwent a mastectomy to remove the primary tumor and affected lymph nodes, followed by 12 weeks of adjuvant chemotherapy and 5 years of endocrine therapy. But 6 years after completing her adjuvant therapy, RaDaR ST detected ctDNA in her blood. A breast MRI came back negative, but her clinical team increased examination frequency to every three months. It took 12 months for imaging to confirm that the cancer had returned, confirming what RaDaR ST had signaled a year earlier.
*Data on file
References
Lipsyc-Sharf M, de Bruin EC, Santos K, et al. Circulating Tumor DNA and Late Recurrence in High-Risk Hormone Receptor-Positive, Human Epidermal Growth Factor Receptor 2-Negative Breast Cancer. J Clin Oncol. 2022;40(22):2408-2419. doi:10.1200/JCO.22.00908